
Creating medical devices is a complex endeavor that requires a blend of innovation and regulatory awareness. Dr. Jane Smith, a leading expert in biomedical engineering, emphasizes, "Innovation in medical devices can change lives, but it must be done responsibly." Her words underline the critical balance between technological advancement and patient safety.
As healthcare evolves, the need for effective and user-friendly medical devices increases. Creating medical devices involves multiple stages, from research to prototyping and testing. Each phase demands expertise and attention to detail. Even small missteps can lead to significant consequences. This highlights the importance of an interdisciplinary approach, engaging engineers, clinicians, and patients alike.
Yet, challenges persist in this industry. The path to market is fraught with regulatory hurdles and financial limitations. Despite advancements, not every idea becomes a successful device. Reflecting on failures can foster growth and innovation. Understanding what did not work is just as important as celebrating successes. In creating medical devices, learning from the past is invaluable for shaping a better future in healthcare.

Identifying healthcare gaps is essential for developing effective medical devices. According to a 2022 report from the World Health Organization, over 5 billion people lack access to essential health services. This staggering figure highlights the pressing need for innovative solutions. Understanding patient and provider needs can illuminate areas where devices may fall short.
For example, pain management presents a significant gap in current treatment options. More than 20% of adults experience chronic pain, but many existing devices fail to address long-term relief. Feedback from healthcare professionals reveals dissatisfaction with current pain management tools. This insight is crucial for designers aiming to create impactful devices.
Moreover, the influx of aging populations complicates healthcare delivery. A report by the Aging Institute notes that individuals over 65 will comprise nearly 20% of the U.S. population by 2030. This demographic shift requires devices tailored for elderly patients, focusing on usability and accessibility. Engaging with healthcare providers and patients can uncover these unmet needs and guide the development of transformative solutions.
| Tip Number | Tip Description | Identified Healthcare Need | Gap in Existing Solutions |
|---|---|---|---|
| 1 | Perform thorough market research | Patient monitoring | Lack of real-time data analysis |
| 2 | Engage healthcare professionals during development | Enhanced usability | Difficult to use interfaces |
| 3 | Identify regulatory requirements early on | Compliance and safety | Prolonged approval processes |
| 4 | Iterate based on user feedback | Patient-centric design | Ignoring end-user feedback |
| 5 | Leverage digital health technologies | Remote patient access | Limited telehealth integration |
| 6 | Focus on affordability | Access to treatment | High cost for patients |
| 7 | Consider interoperability with existing systems | Data sharing | Siloed data practices |
| 8 | Utilize advanced materials | Durability and performance | Short lifespan of devices |
| 9 | Trial prototypes with real patients | Effective testing | Failure to match patient needs |
| 10 | Create a robust support system for users | Ongoing user assistance | Lack of training and resources |
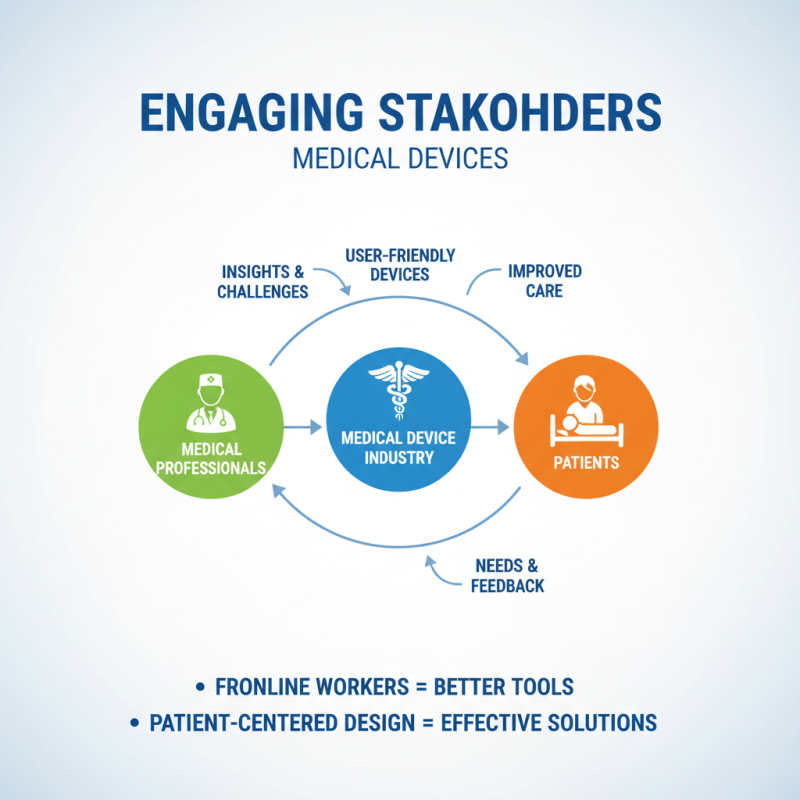
Engaging stakeholders is crucial in the medical device industry. Collaboration with medical professionals is essential for understanding the challenges they face. Frontline workers know what tools can improve patient care. Their insights can guide the design process, ensuring that devices are user-friendly and effective.
Patients also play a vital role in this collaboration. Their experiences with current technologies offer valuable feedback. Listening to their needs can lead to innovations that truly address their concerns. Prototyping or beta testing with patients can yield new ideas for improvement. However, balancing diverse needs can be challenging.
Designing a device is not just about functionality—it’s about empathy. Continuous engagement with both professionals and patients fosters a culture of innovation. This process is ongoing and requires reflection. Questions like “Does this truly help the user?” must be asked repeatedly. Accept that there will be setbacks. Learning from these moments can lead to breakthroughs.
Incorporating user-centric approaches in medical device development is crucial. Start by engaging users early in the design process. Collect direct feedback from healthcare professionals and patients. This ensures that devices meet real-world needs.
Tips for creating effective devices: focus on usability. A complicated interface can lead to user errors. Simplify design and instructions. Conduct usability testing to identify potential issues before launch. Iteration based on user feedback is vital for improvement.
Another important aspect is empathy. Understand the context in which users will operate your device. Users face stress in medical situations. If your device is difficult to use, it adds more anxiety. Designing for ease and clarity can significantly improve user experience. Remember, not every design will be perfect initially. Embrace feedback and be ready to make necessary adjustments.

Navigating regulatory compliance is crucial for medical device development. Understanding FDA regulations and international standards can significantly impact the success of a product. The FDA has specific guidelines that must be followed. This includes design controls and risk management processes. A misstep in these areas can lead to failed approvals.
Many innovators overlook the importance of documentation. Adequate records ensure compliance and facilitate smoother audits. Clear labeling and user instructions are also essential. They help avoid misunderstandings and product misuse. Inaccurate documentation may lead to costly recalls.
Navigating the complexities of global regulations adds another layer. Each country has unique requirements. Companies must invest time in understanding these differences. This can be challenging, but it is necessary. An evident gap in many teams is a lack of regulatory knowledge. Continuous education and training can mitigate this risk. Embracing a proactive approach to compliance not only strengthens product quality but also builds trust within the healthcare system.
Prototyping is crucial in medical device development. It allows teams to visualize concepts early. According to a report by the FDA, 86% of successful devices were tested multiple times before finalization. This iterative approach is vital for refining functionality and usability. It helps uncover design flaws that may not be evident in initial sketches.
Testing varies across stages. Early prototypes focus on basic functionality. Later prototypes incorporate user feedback, addressing real-world challenges. A study from the Medical Device Innovation Consortium highlighted that user-centered design leads to a 50% reduction in product recalls. Iterative testing ensures that devices meet the needs of healthcare professionals and patients alike. It brings about a necessary collaboration between engineers, designers, and clinicians.
Facing flaws is part of the process. Some prototypes may fail to meet expectations in critical areas. This is an opportunity for reflection and improvement. Embracing failure can lead to innovations that genuinely transform healthcare. Practical evaluation creates resilience in product design. Effective prototyping and testing are foundational for meaningful advancements in medical devices. Ensuring optimal performance isn't just a goal—it's an ongoing journey.




|
This is a medical device. |
