
A medical device plays a crucial role in modern healthcare. These devices range from simple tools like thermometers to advanced machinery like MRI machines. Understanding how a medical device functions can illuminate its importance in diagnostics and treatment.
Medical devices operate using a combination of mechanics, electronics, and software. For instance, a pacemaker regulates heart rhythm through electrical impulses. This example highlights the intricacies of these devices. They are designed to improve patient outcomes and provide essential information for healthcare providers.
Despite their benefits, medical devices can sometimes fall short. Issues can arise from malfunctions or inadequate user training. These challenges remind us that continuous improvement is vital in healthcare technology. Ensuring reliability in a medical device is essential for patient safety and effective treatment.
Medical devices play a crucial role in modern healthcare. They range from simple tools like thermometers to advanced imaging machines. According to a report by GlobalData, the global medical device market is expected to reach over $600 billion by 2024. The growth highlights their increasing importance in diagnosing, monitoring, and treating various health conditions.
The functionality of medical devices is diverse. They can enhance patient care and streamline procedures. For example, wearable devices track vital signs continuously. These devices contribute to preventive medicine by identifying potential health issues early. Yet, the industry faces challenges like regulatory hurdles and the need for innovation. Some devices may not function as intended in all patient populations, raising concerns about their effectiveness.
Healthcare professionals must be well-versed in selecting and using these devices. Training is often incomplete, leading to misuse or underutilization. A study published in the Journal of Medical Devices noted that nearly 30% of healthcare workers reported inadequate training. This gap showcases a need for ongoing education. Overall, while medical devices are essential, their implementation requires careful consideration and improvement.
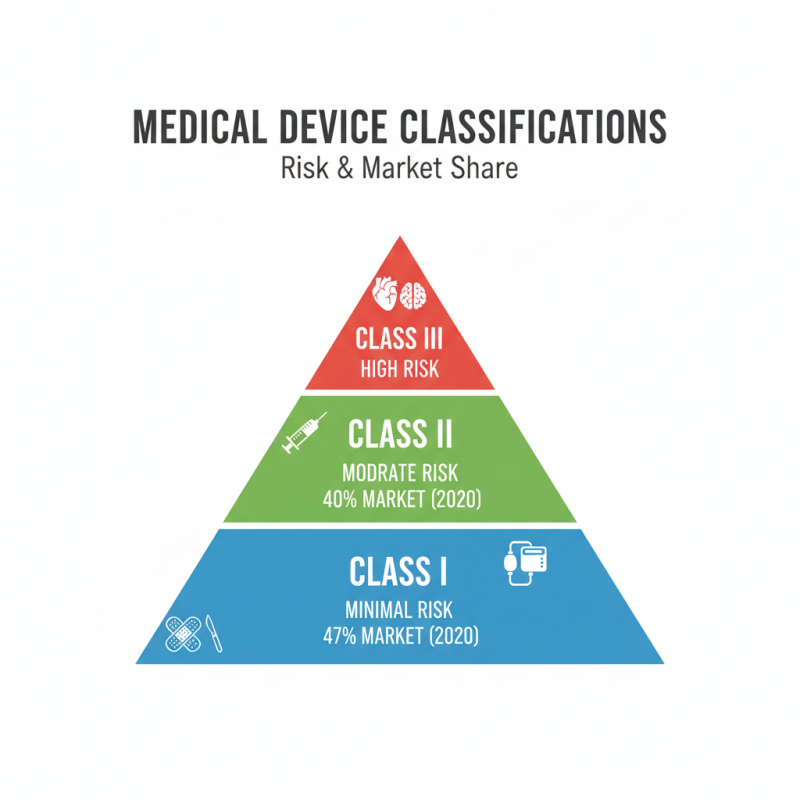
Medical devices vary widely in terms of their purpose and associated risks. They can be classified into three primary categories: Class I, Class II, and Class III. Class I devices typically pose minimal risk. Examples include bandages and handheld surgical instruments. These devices usually require the least regulatory control. In 2020, it was estimated that Class I devices accounted for about 47% of the global medical device market.
Class II devices present a moderate risk. They include items such as infusion pumps and diagnostic imaging systems. Regulatory control is tighter for these devices because they must meet specific performance standards. Reports suggest that Class II devices made up approximately 43% of the market. This highlights their critical role in patient care and the need for ongoing evaluation of their safety.
Class III devices are the highest risk category. Examples are implantable devices like pacemakers. They necessitate pre-market approval due to potentially life-threatening repercussions if they malfunction. In 2021, Class III devices represented around 10% of the market but had significant regulatory demands. The complexities in monitoring and ensuring quality in this category require continued reflection and improvement from manufacturers.
Medical devices come in various forms. They can range from simple tools such as thermometers to complex machines like MRI systems. Each device has specific components that contribute to its overall functionality. Understanding these components is key to grasping how a medical device works.
A typical medical device usually consists of a sensor, a processor, and an output mechanism. The sensor gathers data from the body. For instance, glucose monitors detect sugar levels through the skin. The processor analyzes this data and sends signals to an output mechanism, which may provide visual or auditory feedback. Sometimes, devices malfunction or provide inaccurate readings. These flaws highlight the need for continuous quality checks and improvements in design.
Additionally, power sources are crucial. Many devices rely on batteries or electricity to function. Yet, power failures can compromise operations. It’s essential to consider how devices can remain reliable in emergencies. Reflection on these challenges can drive innovations in medical device technology. Understanding both the capabilities and limitations fosters better health outcomes.
This chart illustrates the distribution of key components found in various medical devices, highlighting their functional roles and importance.
Regulatory standards for medical devices are crucial. They ensure safety and effectiveness for users. Various agencies worldwide enforce these standards. These typically involve rigorous testing and evaluation. Before a device reaches the market, it must undergo several stages.
The approval process starts with pre-market submissions. Manufacturers must provide extensive documentation. This includes clinical trial results, safety data, and design specifications. Review timelines can vary significantly. Some devices may take months, while others could take years. It’s a meticulous process, but sometimes efficiency is questioned.
Once a device is approved, post-market surveillance becomes essential. This monitoring can reveal unforeseen issues. Some devices may encounter problems only after widespread use. A considerable challenge remains: ongoing compliance with regulatory updates. Companies must adapt quickly to keep up. These dynamics highlight the industry's need for constant vigilance and improvement.
The future of medical device innovation is vibrant, fueled by advancements in technology. According to a recent report by Deloitte, the global medical device market is projected to reach $612 billion by 2025. This growth is partly driven by the integration of artificial intelligence (AI) and machine learning into devices. These technologies enhance diagnostics and patient monitoring. However, the rapid pace of innovation can outstrip regulatory processes. This mismatch often raises questions about safety and efficacy.
Telemedicine is another trend transforming medical devices. A study by the American Medical Association showed a 154% increase in telehealth visits. Devices that enable remote patient monitoring are crucial here. They offer real-time data to healthcare providers. Despite the benefits, concerns about data privacy persist. Many devices collect sensitive information. The challenge lies in balancing innovation with security and ethical considerations.
Moreover, wearable health technology is gaining traction. A report from Grand View Research suggested that the wearable medical device market could surpass $27 billion by 2025. While wearables provide valuable insights into health, they may create over-reliance on technology. Users may misinterpret data without proper guidance. The future of medical devices will require ongoing dialogue about benefits and risks.
|
This is a medical device. |
